|
Preserve in a dark coloured bottle.(6.8g/l)ĭissolve 44 g of the salt in 1 liter of water. Saturate 250ml of water with chlorine.the chlorine may be prepared by dropping conc. Shake 3 g of the salt with 1 liter of water, filter and decant the saturatrd solution after several hours (0.015M) Add more bromine if necessary to a slight excess.ĭissolve 55 g of the hydrated salt in 1 liter of water (0.25M) (1M)ĭissolve 71 g of the crystaline salt in 1 liter of water.(0.5M)ĭissolve 132 g of the salt in 1 liter of water.(1M)ĭissolve38 g of the salt in 1 liter of water (0.5M)ĭissolve 61 g of the salt in 1liter of water.(0.25M)Ī saturated aqueous solution is prepared by shaking 35g or 11ml liquid bromine with water. (2M)ĭissolve 169 g of the salt in 1 liter of solution (3M).ĭissolve 80 g of the salt in 1 liter of water. Dissolve 192 g of the salt in a mixture of 140ml conc.Īmmonia solution and water to make up 1 liter of solution. 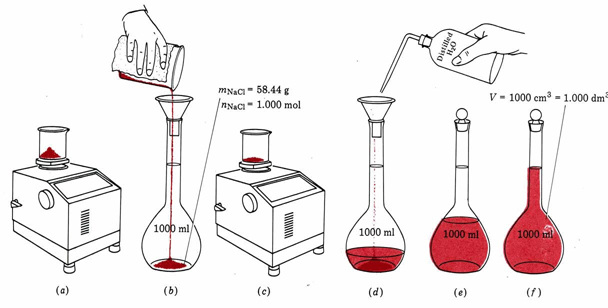
The commercial salt is a mixture of bicarbonateĪnd carbamate. If reagent not found in this list check the alphabetical listing below.ĭissolve 231 g of the salt in 1 liter of water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
